The Reuters Events’ Pharma Awards focus on what really matters: value for patients and customers. Recognising innovative and meaningful approaches, this is the work worth applauding. Let’s deliver outcomes, restore pharma’s reputation and celebrate real achievements.
We’re delighted to announce the following companies have been shortlisted:
Look through our various award categories by browsing through the tabs below.
Patient Champion Awards
Patient Champion Award: Patient Advocate
The winner of this award will be a person or small group which identifies as a patient (or patient group), and who has provided other patients or people with a stronger voice, through advocacy work. This voice will have been influential enough to have created communities, changed policy, raised awareness, helped launch a product or driven a new movement.
-
 Submission Title: Julie CronerRead More
Submission Title: Julie CronerRead MoreAfter graduating from West Virginia University, Julie worked at Deloitte Consulting for 5 years. As a Senior Technology Integration Consultant, she specialized in all areas of the software development lifecycle, successfully leading teams through the planning, design and implementation of new systems.
As she traveled the world and continued to climb the ladder as a female consultant in tech, her time was cut short with the diagnoses of several chronic and debilitating health issues. Ultimately, Julie was forced to take disability leave for three and a half years.
Through doctors’ appointments, procedures, and life-altering changes such as losing the ability to walk, Julie refused to sit back. She started a blog (itsjustabadday.com) to share her experience with chronic illness, went back to school and received her certification in Holistic Health Coaching. Most importantly, she dedicated the majority of her time to patient advocacy work.
As her passion for advocacy grew, so did her reach, earning her speaking engagements with the FDA and at conferences such as Stanford Medicine X, ePharma Summit and many other venues. Advocacy work became her full-time career.
Eventually, Julie decided to choose a new career path centered around her advocacy work and joined WEGO Health, now Health Union, as Vice President of the Patient Leader Network. In her role, she launched the world’s first online platform dedicated to Patient Leaders, which offers resources and training, as well as paid collaboration opportunities between healthcare companies and patients passionate about building a more patient-centered healthcare system.
As Julie says, “Leading the WEGO Patient Leader Network has proven to be one of the most rewarding pieces of work I've ever had the chance to do. Every day I work with the MOST inspiring Patient Leaders to collaborate and push forward a more patient-centered healthcare system.”
When Julie isn’t fighting to ensure the patient voice is heard, she can be found running after her two (soon to be three) toddlers, with her husband Josh, in Pittsburgh, Pa.
-
 Submission Title: The UK Sepsis TrustRead More
Submission Title: The UK Sepsis TrustRead MoreThe threat and impact of COVID-19 is obvious, with millions of lives claimed and irreputable damage to health systems, economies and individual wellbeing across the globe. However, one of the less-talked about aspects of COVID-19 are the blurred lines between its symptoms and after-effects, and those of sepsis.
Sepsis is the immune system’s overreaction to an infection or injury. If not treated quickly, sepsis can result in organ failure and death. Yet with early diagnosis, it can often be treated.
Globally, sepsis is the cause of 11 million deaths a year, accounting for 1 in 5 of all lives lost. Nationally, 245,000 people suffer from sepsis, with 48,000 dying every year, 9,000 more than the combined fatalities of bowel, prostate and breast cancer.
COVID-19, like any infection, can lead to sepsis and it’s estimated that at least 25% of COVID-19-related deaths were due to sepsis. Moreover, people who have survived COVID-19 are likely to have an increased risk of developing sepsis during their recovery.
Due to the similarities between its symptoms and those of COVID-19, sepsis can be difficult to detect – both trigger a disproportionate immune response, as well as sharing symptoms such as fever spikes, breathlessness and muscle pain. However, in 2020, with the pandemic in full swing, these similarities were little understood, with thousands needlessly suffering as a result.
Aware that many lives could be saved, The UK Sepsis Trust (UKST) launched a health communications campaign entitled ‘Blurred Lines’.
The campaign aimed to spread the message to patients and their families, offering a simple instruction – “Just ask: could it be sepsis?”.
The campaign had a significant impact, cutting through the noise of the pandemic to reach patients and their loved ones.
The results surpassed all expectations, with 40% of patients supported by UKST now COVID-19 related.
UKST aims to make people aware of both conditions and the signs to look out for, and to feel empowered to act fast if they suspect sepsis. In short, the charity exists to fight sepsis, stop preventable deaths and support those affected by these two conditions.
-
 Submission Title: Mental Health Screeners & Resources to Educate, Support and Intervene Expecting and New Moms With An Elevated Risk of PPDRead More
Submission Title: Mental Health Screeners & Resources to Educate, Support and Intervene Expecting and New Moms With An Elevated Risk of PPDRead MoreDetails coming soon.
-
 Submission Title: Seth RotbergRead More
Submission Title: Seth RotbergRead MoreFrom an early age, Seth faced adversity when learning about his mother’s diagnosis of Huntington’s Disease (HD) at the age of 15. Five years later, Seth went through genetic testing and found out he tested positive for HD and may end up like his mom one day. Seth is more than just a patient advocate – he helps make a difference in the community by immersing himself in the patient perspective. His own health journey has provided many years of hands-on experience in fundraising, patient advocacy, volunteering, B2C content, strategic planning, event planning, and program management. Seth loves to connect with people and build relationships across all stakeholders within the health space; ranging from life science companies to local patient advocacy organizations. He has over 12 years of nonprofit leadership experience, including being the co-founder of Our Odyssey.
-
 Submission Title: Ilana JacquelineRead More
Submission Title: Ilana JacquelineRead MoreIlana Jacqueline is a speaker, author, advocate, and healthcare marketing professional. She is the author of the book Surviving and Thriving with an Invisible Chronic Illness (New Harbinger, 2018). Her advocacy has spanned all segments of the industry including non-profits through her role as Managing Editor at Global Genes, patient data collaborations through her work as Director of Patient Advocacy and creator of the Genomics Collaborative at FDNA, INC. and patient education through her column at IG Living Magazine. She currently works as Associate Director of Community at Health Union/ WEGO Health. She is enthusiastic about new media, short form video and social strategy to help in bridging the gap between patients and the power of collaboration with pharma and healthcare brands. She has openly shared her story of living with Primary Immune Deficiency Disease, a rare disease, which inspired the premise for the Lifetime show “Behind the Mystery: Rare and Genetic” produced by Carri Levy and Molly Mager. Ilana has sat on the board of multiple rare disease non-profits and is eager to help young advocates aspiring to use their personal experience with chronic illness and their professional expertise to make a difference.
Patient Champion Award: Pharma
The winner of this award will be an individual or small group which has demonstrably worked to make patient’s lives easier from within a pharmaceutical company, perhaps (but not necessarily) going against the grain of commercial interests, or forging a new path in order to champion or deliver a patient cause.
-
 Submission Title: Partnering With a Community Advisory Board to Build DE&I into Clinical ResearchRead More
Submission Title: Partnering With a Community Advisory Board to Build DE&I into Clinical ResearchRead MoreLack of awareness, lack of education and lack of access to clinical trials have presented significant barriers to participation for members of underrepresented communities. These issues are further compounded by mistrust due to historical events and injustices in past clinical trials. In order to enroll more diverse representation in clinical trials with the hope of developing better medicines for all, Biogen acknowledged the need to form a partnership with underrepresented communities to address some of these barriers.
Biogen partnered with the Center for Information on Clinical Research Participation (CISCRP), a nonprofit dedicated to educating patients and the public on clinical research participation, to build a community advisory board (CAB) among patient advocates identifying as Black/African American and/or Hispanic/Latino.
The CAB was assembled, in part, to help educate Biogen team members on central issues regarding race, ethnicity, associated health disparities, and educational/awareness initiatives. The primary goal of Biogen and CISCRP was to listen and gather as much insightful feedback as possible from the CAB. Specifically, the CAB was designed to provide feedback on clinical trial-related patient-facing materials and designs. These insights directly impacted Biogen’s patient-facing materials and website content to create engaging, educational tools to increase awareness and and participation from these communities in clinical trials. Insights from this group have also helped to shape the future objectives and strategies of the UP initiative and have helped to educate Biogen and CISCRP on issues of diversity and inclusion in clinical trials more broadly.
Furthermore, the CAB’s willingness to share their lived experiences provided a depth of understanding on issues of health equity which would not have been possible without their insight. While the impact they have had on Biogen’s patient-facing content has been substantial, with hundreds of actionable pieces of feedback shaping material development, the CAB has also had a fundamental and transformational impact on CISCRP’s and Biogen’s understanding of health equity issues.
Additionally, because of the value and success demonstrated by the current CAB, we plan to continue the partnership and expand the CAB to further include the voices of a broader range of underrepresented populations, races and ethnicities.
-
 Submission Title: Patient Expert CouncilRead More
Submission Title: Patient Expert CouncilRead MoreTo Servier Pharmaceuticals, a patient’s voice is one of the most important and treasured assets a healthcare company can have. In fact, as a patient-first organization, Servier believes strongly that the opportunity to hear patient expert input is a privilege and the foundation for guiding strategy. Comprised of patients, caregivers, social workers and other experts, Servier Pharmaceuticals created the Patient Expert Council to ensure that the patient expert input was at the core of the company. This is, in many ways, what patient advocacy is all about – not simply listening to, but also making an active effort to include patient voices into every aspect. The Council now serves as a comprehensive, one-of-a-kind resource that integrates the patient perspective and voice into every step of the decision-making process across all functions within the organization.
This past year, against the backdrop of the global COVID-19 pandemic, the role of the Patient Expert Council has risen to new heights, ensuring that patients are adequately supported during this unprecedented time. Servier Pharmaceutical’s deliberate and unwavering patient focus has uniquely positioned the company to quickly adapt to the new needs patients were facing and provide value in new and innovative ways. Patient expert insight was even more important due to the pandemic and the Council immediately sought ways to help patients’ new needs across all aspects of the business, from developing new digital tools, expanding into new therapeutic areas, to patient facing programs.
Servier has always seen its patient experts as partners. While it’s important to create innovative medicines that help save patients’ lives, it’s equally important to partner with patient experts, incorporate their insights during the development and creation of programming and jointly build the tools, resources and support needed to navigate treatment and survivorship. The Patient Expert Council is a distinctive offering that allows patients to advocate for and assist those going through a similar experience as they have. Through the Patient Expert Council and more, Servier Pharmaceuticals is leaving a lasting legacy of patient centricity, ingenuity and innovation – working with patients to help bring them the promise of tomorrow.
-
 Submission Title: Diversity, Equity, and Inclusion in Clinical TrialsRead More
Submission Title: Diversity, Equity, and Inclusion in Clinical TrialsRead More“That big red container sitting in the corner of my room is a constant reminder of my disease”
-Patient Insight
In 2019, the Janssen Supply Chain (JSC) Customer Focused Innovation team assumed leadership of the Janssen Safe Returns® Program. The team saw this as an opportunity to redesign the program that offered Janssen customers in the US safe and convenient disposal of their self-injectable devices.
The JSC team pulled together a group of experts to look at how the program could drive impact for patients. As the team worked to update the value proposition, it realized that it was critical to understand patient needs.
The team followed an agile innovation approach:
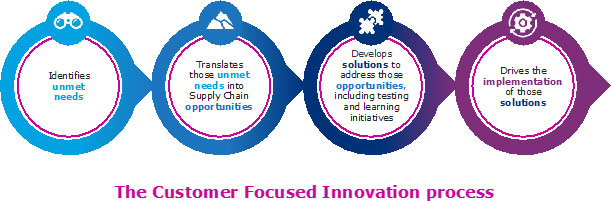
Intensive market research with patients led to insights related to convenience, sustainability, and design improvements. Design workshops were conducted to create a service blueprint for the new program.The new service blueprint focused on:
a. Convenience: The container design was changed from a bulk plastic container that needed to be taken to the post office to a single-use envelope that the patient can leave in their mailbox.
b. Automated replacement: An automated replacement process was created to ensure that patients would receive the return envelopes in time.
c. Environmental impact: The single use return envelope was designed to be more environmentally friendly by using new material called ‘power paper’.
d. Simple: The team worked to ensure that the information related to program was made available to the patients through existing connection points.
e. Adherence: Potential to have positive impact on patient adherence due to increased engagement and frequency of return being linked to frequency of dosing.The updated Safe Returns® Program has many benefits
1. Increased patient engagement: from every 2-4 years to every 4-8 weeks
2. Patient participation rates increased by 300% within the first few weeks
3. The new program has potential to reduce carbon footprint of the program by ~300%
4. The updated systems provide JSC insights on patient needs and experience which enables the team to remain patient focusedThe success of the program has led to its adoption in broader patient engagement initiatives, as well as launch of the program in global markets.
-
 Submission Title: Safe Returns® Program Innovation for Patient ValueRead More
Submission Title: Safe Returns® Program Innovation for Patient ValueRead MoreThe Janssen Pharmaceutical Companies of Johnson & Johnson is focused on delivering transformational medicines to patients in need. As part of this mission, the Global Development team is striving to reshape the clinical trial process for all stakeholders and, most importantly, patients.
To help advance this mission, the Diversity, Equity, and Inclusion (DE&I) in Clinical Trials team was formed as a working group to develop and execute upon strategies for ensuring that clinical trials are more diverse, equitable, and inclusive across therapeutic areas so that all patients can benefit from transformative medicines effectively and safely.
This strategy includes:
•Culture – Nurturing an internal culture that expects and promotes behaviors that support DE&I approaches in clinical research as standard practice.
•Access – Applying strategic and operational practices that address and reduce systemic and tactical barriers to access.
•Awareness & Trust – Leveraging our unique role as leaders to facilitate ongoing collaborations and transparent conversations that help improve awareness and trust within the communities we serve.The need for greater equity in healthcare was only intensified by the COVID-19 pandemic. With this urgency in mind, we created the Research Includes Me Community Engagement and Education Program, focused on reaching underrepresented communities by meeting them where they are - on the ground and through transformative technologies, partnering with trusted voices, and addressing barriers to participation in clinical trials. Specifically, we:
•Created the website ResearchIncludesMe.com to provide information on the clinical research process, why diversity and inclusion is so important, and offer resources related to clinical trials. We have seen significant traffic on the site, suggesting that communities are benefiting from the information.
•Partnered with U.S. Mobile Health during unprecedented times to coordinate and implement engagements within populations across the Southeast U.S. that were disproportionately affected by the COVID-19 pandemic. The actions are showcased in a video, demonstrating how education helped alleviate existing concerns and establish trust to further drive participation. -
 Submission Title: Diverse Representation in Clinical TrialsRead More
Submission Title: Diverse Representation in Clinical TrialsRead MorePeople respond differently to medicines based on their age, sex, race, ethnicity and other factors, making diverse representation in clinical trials critical, yet many minority communities are underrepresented. Lilly has been working to address this industry-wide problem for more than a decade, and the onset of the global pandemic amplified existing health disparities and their impact on research.
Lilly is intensifying its efforts to better understand and represent Black Americans as well as other minority populations by:
• Creating a robust clinical trial strategy to reach diverse populations;
• Selecting a diverse range of trial sites and investigators; and
• Increasing diverse representation through collaborations.Through collaborative efforts, Lilly is improving our ability to enhance representation of Black women in our breast cancer clinical trials. Working with advocacy groups including The Breast of Us and Tigerlily Foundation, we’re assessing protocol elements to maximize participation for women of color and leverage decentralized capabilities within the trial. We are also partnering to educate and reach patients where they are with support resources. In addition, we located study sites in areas of the U.S. with higher prevalence rates of black women with breast cancer.
Another aspect of our efforts is enhancing site-facing technology to better understand the race/ethnicity of patients in prescreening and rationale behind screen failures. This data will help Lilly improve clinical trial recruitment for women of color living with breast cancer, and make it easier for them to participate in research by adapting inclusion and exclusion criteria, reducing in-person visits and revising data collection methods.
Additionally, we are taking steps to enhance investigator diversity by working through partners to recruit more oncologists of color as investigators on our trials. Historically, race/ethnicity data hasn’t been collected for investigators, so we have introduced a survey to self-report this information and measure progress towards our goals.
With a strong investment in understanding diverse patients, through technology, data and partnerships Lilly is working to establish new standards for increasing trial participation. We have much work to do in this area and are excited to build on these efforts, to ensure our medicines are safe and effective for all those who need them.
"Most Valuable" Awards
Most Valuable HCP Initiative
The winner of this award will be an innovative project which has delivered tangible working and healthcare improvements, both in the long and short term, to healthcare professionals or to healthcare institutions/systems.
-
 Submission Title: CHIEF-HF: reimagining clinical trials for the futureRead More
Submission Title: CHIEF-HF: reimagining clinical trials for the futureRead MoreCHIEF-HF is Janssen’s first-ever fully decentralized, randomized, interventional clinical trial. The trial deploys a novel method, re-imagining clinical trial design to accelerate research while prioritizing patient safety and convenience.
Cardiovascular disease remains the leading cause of death in the U.S., and widespread concerns have arisen about the costs, inefficiencies, and complexities of clinical trials. Against this backdrop, Janssen designed the CHIEF-HF trial, which directly engages participants without in-person contact. Data is collected directly from participants via a phone app, a wearable device and insurance claims. Trial participants are supported by a virtual PI within their health network and a virtual coordinating center. An unplanned benefit of the trial design was its relevance and feasibility during the COVID-19 pandemic, which had major disruptive consequences on traditional clinical trials. The study was designed by Janssen Scientific Affairs, LLC, with an Executive Steering committee, and PRA Health Sciences.
In addition to prioritizing patient convenience, Janssen is committed to ensuring our clinical trials ease burdens to participation for historically underrepresented communities. The design and operational execution of CHIEF-HF, driven by innovation and forward thinking, include a deliberate approach to enable recruitment of a diverse participant group.
We know that to develop more effective, life-changing therapies faster, we must continue to challenge the status quo and reimagine how we conduct clinical trials today. By bringing cutting-edge science, creative approaches, and innovative technologies together, Janssen believes we can create more inclusive clinical trials that deliver an understanding of which drugs and treatments have the best results for all patients.
-
 Submission Title: The AMAZE Disease Management PlatformRead More
Submission Title: The AMAZE Disease Management PlatformRead MoreDetails coming soon.
-
 Submission Title: Medtronic PVI Occlusion AppRead More
Submission Title: Medtronic PVI Occlusion AppRead MoreMedtronic partnered with Tipping Point Media to enhance their current methods for training health care providers and sales representatives in the proper usage of the Arctic Front™ family of cryoablation catheters and various techniques for how to properly perform the cryoablation procedure to treat atrial fibrillation.
The Medtronic team identified two major issues with their previous training techniques, which involved setting up a silicone model of a heart and having HCPs and sales representatives meet to perform the training in-person.
First, the silicone heart model is static and did not respond to the steps of the procedure as a live heart would during a real procedure. Learners were learning the step-by-step process but not gaining key decision-making skills. In addition, the silicone heart model is fixed to a standardized anatomy, meaning learners could not practice how to perform the procedure on abnormal anatomy or unique cases.
Second, the silicone model training was cost-prohibitive and cumbersome to transport, schedule, and set-up. Having a digital training solution that was easy to deploy and access would increase scalability and translate to a greater consistency in language, terminology, and best practices, which would improve patient safety.
-
 Submission Title: ‘Hands-on’ Technical Training in a Virtual WorldRead More
Submission Title: ‘Hands-on’ Technical Training in a Virtual WorldRead MoreNecessity drives innovation, engaging us to rethink existing paradigms and leverage new opportunities. Our HCP focused initiative showcases exactly that, highlighting the provision of technical medical skills training (in this case, the insertion and removal of a subdermal contraceptive implant to healthcare professionals (HCPs) across Canada), amidst a global pandemic and nationwide lockdowns.
In this context, we developed a virtual technical skills training platform, with a focus on simulation-based insertion and removal procedures, conducted on a model arm. We were able to effectively translate a traditional face-to-face experience to a virtual setting, with a digital solution that could rapidly scale-up to meet the anticipated volume of training needs.
To this end, a key innovation was the development of a single-source web platform for training, providing HCPs with clear, simple guidance to access training and integrating pre-requisite online training content, as a precursor to registering for a ‘hands-on’ practical training event. All these elements were delivered as a seamless ‘end-to-end’ user experience for HCPs, while meeting their training needs.
The scale and impact of this initiative has been notable: over 2200 HCPs trained since September 2020, including physicians, nurse-practitioners and nurses across all provinces and territories in Canada. Beyond numbers, we’ve witnessed an unprecedented level of HCP engagement: of the two-thirds who completed post-training evaluation forms, over 97% of respondents validated that the virtual format met its objective. Our approach also allowed us to have much broader HCP reach (and so impact), than would have been possible face-to-face, especially in context of resource limitations.
Advancements in virtual medical (especially technical) training have the potential to facilitate wider access to knowledge, not only in gynecology, but other medical disciplines as well, satisfying two important needs: 1) an ‘active’ and enriched experience to learn; and 2) a remote education experience that reduces geographical and resource-based constraints, particularly in a vast and not densely populated country such as Canada. Whilst the COVID-19 pandemic may have been the catalyst, the ‘pearls’ gained in this endeavor can be more broadly applied across fields of medicine.
-
 Submission Title: Institute of Leadership ExcellenceRead More
Submission Title: Institute of Leadership ExcellenceRead MoreAt Janssen, we believe that to develop more effective, life-changing therapies faster, we must continue to challenge the status quo. We will apply insights from CHIEF-HF to make early informed decisions in an effort to transform future trial design and improve the trial experience for all patients and sites we serve.
Traits such as Emotional Expressiveness, Self-Confidence, Self-Determination, Freedom from Internal Conflict and Communication Skills are crucial for any leader, especially Healthcare Leaders. Undoubtedly, the Healthcare professionals (HCPs) of Pakistan are some of the most technically skilled among the world. However, they lack the necessary soft skills and the above-mentioned traits for these are not taught academically, or incorporated into the HCP’s on-job training.
As part of our vision to promote “A Healthier Society”, PharmEvo stepped in to bridge this gap in healthcare management. We launched the Institute of Leadership Excellence (ILE), a CSR initiative to develop healthcare leaders who could work to improve overall healthcare delivery. Our focus was on 3 key areas:
1. Developing leadership Skills: Through programs such as Grid Leadership Model, 7 habits of highly effective people program, Speak like a Leader program etc.
2. Strategy Formulation & Development: Through trainings on identifying purpose, setting vision/mission etc.
3. Execution of Strategy: Through different training modules on learning to execute the developed strategy.ILE became the first platform in Pakistan to be certified by the Franklin Covey institute for ‘The 7 Habits of Highly Effective People’ training for healthcare professionals. ILE has a Leaders Club for healthcare executives that provides content for learning and intellectual engagement. We also collaborated with Talisium to provide leadership courses tailored for healthcare professionals from prestigious institutes such as Royal College of Physicians (UK), NHS Academy of Leaders (UK) and others.
These efforts have resulted in improvement of health outcomes, improvement in the quality of healthcare services, enhanced patient safety, and cost efficiency.
ILE has received recognition from international management and marketing gurus. Recently ILE was included as a case study in Philip Kotler’s upcoming book “Essentials of Modern Marketing”.
ILE plans to establish Centers of Healthcare Leadership (CoHLs) in 100 institutes across Pakistan in the next 5 years, and by 2026 employing a sizeable number of training personnel for these CoHLs.
Most Valuable Collaboration
The winner of this award will be the creator of a pharma-pharma or pharma-nonpharma initiative which clearly brings a new proposition to either HCPs, payers and/or healthcare systems, creates new value and redefines what is possible in our industry with a degree of lateral thinking.
-
 Submission Title: Pfizer and the Historic Biopharma Industry Pledge to #StandWithScience on Coronavirus VaccinesRead More
Submission Title: Pfizer and the Historic Biopharma Industry Pledge to #StandWithScience on Coronavirus VaccinesRead MoreWhen the World Health Organization declared a COVID-19 pandemic on 3/11/20, the best and brightest minds globally set out to do the impossible – create a safe, effective vaccine that would reverse the spread of COVID-19. Leading that charge were Pfizer and BioNTech, which launched Phase I clinical trials on a COVID-19 vaccine in mid-March 2020.
National polls in August/September 2020 unearthed widespread vaccine skepticism and ranked the pharmaceutical industry as the most poorly regarded – even lower than the Federal government, which had drawn heavy criticism for its pandemic response. 62% of Americans believed that political pressure would force the FDA to rush approval of an unproven vaccine. Only 21% of voters said that they would seek vaccination as soon as a vaccine became available.
Pfizer set out to transform this perception by increasing public confidence that the scientific and regulatory process for the COVID-19 vaccine would be held to the highest standards, establishing trust and goodwill and overcoming misinformation that had tainted the public’s view of Pfizer’s motivations to find a solution for COVID-19.
On 9/8/20, nine CEOs of companies developing vaccines signed the historic Pledge, committing to uphold the integrity of the scientific process and make the safety and well-being of vaccinated individuals their top priority. United under #WeStandWithScience, Pfizer began the virtual handshake on Twitter, then tagged GlaxoSmithKline to join the Pledge, which tagged J&J and so on to establish a bold, multi-Tweet movement. Each company followed up with individualized social posts on Twitter and other social channels.
The #WeStandWithScience Pledge resulted in one of the biggest news days in Pfizer history, garnering blanket top-tier coverage. Pfizer was at the forefront of the conversation and saw positive coverage from virtually every top-tier media outlet, resulting in a 12.8% increase in positive sentiment and a nearly 40% increase in share of voice compared to the 2019 baseline.
Americans feared a vaccine tainted by politics and corporate greed. By assuaging those concerns through transparency and a publicly affirmed commitment to science, Pfizer continued to increase vaccine confidence.
-
 Submission Title: Roche and Regeneron Collaborate to Significantly Increase Global Supply of Investigational Antibody Combination REGEN-COV for COVID-19Read More
Submission Title: Roche and Regeneron Collaborate to Significantly Increase Global Supply of Investigational Antibody Combination REGEN-COV for COVID-19Read MoreIn August 2020, Roche and Regeneron announced that we were joining forces in the fight against COVID-19 to develop, manufacture and distribute REGEN-COV (casirivimab and imdevimab), Regeneron’s investigational antiviral antibody cocktail, in order to bring the therapy to as many people as possible. Through this collaboration, we expect to increase the global supply of the antibody cocktail to at least 3.5 times of the original capacity and ensure access to more people globally. Regeneron and Roche are responsible for the distribution and manufacturing in and outside the U.S., respectively.
Technology transfers started swiftly and were completed in record time in 2020, enabling Roche to start producing REGEN-COV at its sites. Teams including technical operations, strategy, quality, access and legal were mobilized to support this herculean effort. Positive safety and efficacy data from three Phase 3 studies have proven the effectiveness of the therapy in outpatient, prevention and hospitalization settings. It is also proving to be effective against the emerging COVID-19 variants and will continue to play an important role alongside vaccines. Our partnership underscores what we can accomplish together when we focus our minds on delivering for patients and work as one united community to tackle global public health challenges. It has only been 10 months since our partnership started and we have already delivered 2 million doses (through June 2021) of the therapy to more than 20 countries for emergency use authorization.
This was only possible because everyone at Roche and Regeneron involved in this project collaborated and supported each other on this journey. From manufacturing colleagues who worked overtime, additional shifts and through holidays to colleagues from other functions who volunteered to support shop floor colleagues to speed up packaging work and help in other ways, to medical affairs and policy colleagues liaising with health authorities to accelerate emergency use approvals, everyone worked tirelessly to ensure that we met our commitments to deliver these life-savings therapies to patients in the fastest possible time. Our continued collaboration will ensure that more people, including the most vulnerable, get immediate protection and/or treatment for existing and emerging variants of the virus.
The collaboration being discussed in this submission is one between OneThree Biotech and Jubilant Therapeutics that focuses on the identification of specific biomarkers for new indications in targeted oncology patient populations.
-
 Submission Title: NCD AcademyRead More
Submission Title: NCD AcademyRead MoreEach year noncommunicable diseases (NCDs) kill 41 million people worldwide, and NCDs account for seven of the top ten leading causes of death globally. Four major disease groups (i.e., cardiovascular disease [CVD], cancer, chronic respiratory diseases, and diabetes) account for over 80% of all premature NCD deaths. However, NCDs can often be prevented through education and regular screening.
Through our partnership projects, Viatris is working to foster progress toward achieving United Nations (UN) Sustainable Development Goals (SDGs). The SDG target 3.4 aims to reduce premature mortality from NCDs in 2030 by 33% compared with 2015 levels through prevention and treatment strategies and the promotion of mental health and wellbeing.
Together with NCD Alliance we conducted research to understand the role of primary care health workers in combating NCDs and to understand the gaps and potential solutions to optimize their efforts. Optimize Multi-Disciplinary Care Teams and Leverage Digital Tools were identified as solutions and initiated a partnership with NCD Alliance, World Heart Federation (WHF) and the American College of Cardiology (ACC) to develop the NCD Academy.
The NCD Academy is an interactive, web-based educational platform for healthcare providers worldwide to improve the prevention and treatment of NCDs based on a patient centric approach, acknowledging the multiple comorbidities that exists in many people living with NCDs. The courses on the platform are free, CME-accredited and have been translated into a variety of languages.
The NCD Academy partners with key global scientific societies covering major therapeutic areas, and will have released courses on all ‘big five’ NCDs (CVD, cancer, COPD, mental health and diabetes) by the end of 2021. Three courses have been released including CVD by ACC, oncology in partnership with American Society of Clinical Oncology (ASCO) () and a mental health course with World Psychiatry Association (WPA).Viatris is the founding sponsor of NCD Academy. Our current contribution focusses on the implementation of NCD Academy in countries across the globe. Due to our global footprint and extensive network we support the expansion of NCD Academy partners with many regional and local stakeholders to together ensure optimal uptake and value.
-
 Submission Title: OneThree Biotech and Jubliant Therapeutics CollaborationRead More
Submission Title: OneThree Biotech and Jubliant Therapeutics CollaborationRead MoreIn the collaboration, Jubilant sought to determine the mechanism for its potentially first-in-class dual epigenetic inhibitor targeting melanoma, myelodysplastic syndrome (MDS), and acute myeloid leukemia (AML) and select solid tumor subsets. Utilizing the OneThree Biotech platform, they were able to:
- Generate new data to support the dual inhibition (LSD1 and HDAC6) to drive inhibition
- Pinpoint molecular markers that could be used to identify patients who might benefit from the dual inhibition
- Determine additional target indications for future trialsOneThree Biotech is a spinout from the largest precision medicine institute in the United States. We have demonstrated great success in drug discovery by applying our biology focused, AI enabled R&D process with partners such as AZ, SPARC, Oncoceutics, and more. In 2021 we began our internal development programs that focus on oncology targets we identified using our AI Platform and validated in early stage development. We won the Reuters Entrepreneur Award in late 2020 and are excited to speak about the results of a partnership we’ve had since then!
Jubilant Therapeutics, Inc. is a patient-centric biopharmaceutical company advancing potent and selective small molecule modulators to address unmet medical needs in oncology and autoimmune diseases. Its advanced discovery engine integrates structure-based design and computational algorithms to discover and develop novel, precision therapeutics against both first-in-class and validated but intractable targets in genetically-defined patient populations. The Company's entrepreneurial minded leadership and scientific teams strive for speed and efficiency by employing a business model that leverages the proven and synergistic capabilities of Jubilant Life Sciences Limited's value chain and shared services. Jubilant Therapeutics is headquartered in the U.S. and guided by globally renowned key opinion leaders and scientific advisory board members.
-
 Submission Title: Carer Well-Being Index: Recognizing and Providing Support for Unpaid CarersRead More
Submission Title: Carer Well-Being Index: Recognizing and Providing Support for Unpaid CarersRead MoreDetails coming soon.
Most Valuable Breakthrough / Discovery
This award is for an original product, product enhancement or scientific invention which has the potential and, ideally, early traction to provide significant impact on a patient population. The winner will be a genuine pioneer, bringing something novel and meaningful to the life sciences industry.
-
 Submission Title: REGEN-COV™ (casirivimab and imdevimab)Read More
Submission Title: REGEN-COV™ (casirivimab and imdevimab)Read MoreCOVID-19 has had a devastating impact worldwide. In early 2020, Regeneron leapt into action, applying its homegrown technologies and infectious disease expertise to combat COVID-19. Predicting the virus would mutate, Regeneron used a strategic multi-antibody approach to increase the likelihood the treatment would remain potent against future variants.
Within five months, REGEN-COV™ (casirivimab and imdevimab), an innovative antibody cocktail, was ready for clinical trials. It was the first therapy of its kind to be authorized to treat COVID-19. Today, more than 20 countries have granted it emergency or temporary pandemic use authorizations and Japan has fully approved the cocktail. It is also authorized in the US and UK for post-exposure prophylaxis. Notably, REGEN-COV is recommended by the NIH and maintains potency against all variants of concern.
Regeneron strongly believes in the importance of getting vaccinated; but should someone get sick, REGEN-COV is one of the only COVID-19 treatments proven to reduce symptoms and hospitalizations.
As many worldwide remain at risk, either because they are unvaccinated, or are not completely protected by vaccination, REGEN-COV continues to be an important tool to stem the impact of this virus. Regeneron is committed to working with partners – whether government, local hospitals and physicians – to help fight this disease and help prepare for future outbreaks.
-
 Submission Title: ADUHELMRead More
Submission Title: ADUHELMRead MoreOn June 7, 2021, the U.S. Food and Drug Administration granted accelerated approval for ADUHELM™ (aducanumab-avwa) as the first and only Alzheimer’s disease (AD) treatment to address a defining pathology of the disease by reducing amyloid beta plaques in the brain. The approval represents the culmination of years of collaboration between global pharmaceutical companies Biogen and Eisai, each which have worked tirelessly to address significant unmet needs in this space.
For nearly two decades, the medical and scientific communities have searched for a new treatment for AD. And yet, legacy therapeutics have only treated symptoms of the disease – not the disease itself. The approval of ADUHELM represents an inflection point in society’s collective battle against AD. By addressing a defining pathology of the disease, this novel therapy has the potential to help change the way patients are diagnosed and treated.
Throughout the past year – even as other biotech companies gave up on this high-risk area or shifted focus to the evolving COVID-19 pandemic – Biogen and Eisai have worked diligently, and with a global lens, in order create an AD therapy that was not only effective, but also accessible as well. Accordingly, in terms of the structure of their agreement, which was finalized in 2017, Biogen led the development of ADUHELM, while both Biogen and Eisai worked on co-commercializing the treatment so it would be ready for distribution in the event of approval. Through this innovative structure, both companies aimed to combine their complementary, and regionally specific expertise, with the goal of making an immediate, tangible impact in the fight against AD.
ADUHELM is truly the realization of Biogen and Eisai’s decades-long commitment to innovation in AD, and its accelerated approval by the FDA marks the first time a drug candidate for this disease has made it to this point. Thanks to their hard work, ingenuity and unwavering commitment, the Alzheimer’s community finally has reason for new hope.
-
 Submission Title: SH-111Read More
Submission Title: SH-111Read MoreShorla Pharma (“Shorla”) is a specialty pharmaceutical company that develops innovative oncology drug products for women’s and paediatric cancers. Located in Cambridge, Massachusetts and headquartered in Tipperary, Ireland, we focus on indications where existing treatments are limited, in shortage or inadequate for the target population. New therapies for women and children are often overlooked by large pharmaceutical companies. Our goal is to bring to market products that represent a new standard of treatment for these patients. Our first therapy, SH-111, is a nucleoside metabolic inhibitor indicated for the intravenous treatment of patients with T-cell acute lymphoblastic leukemia and T-cell (T-ALL) lymphoblastic lymphoma (TLBL), an aggressive blood and bone marrow cancer which progresses quickly. While most leukemias present in older people, T-cell leukemia is most common among children, adolescents, and young adults. With no alternative source of SH-111 currently approved, the drug supply has had severe shortages. Targeting this unmet clinical need ensures we bring this life saving treatment to the most vulnerable patients. Although all phases of the innovation cycle are crucial, access to treatment for patients have the greatest societal impact and we have prioritised the development of SH-111 to mitigate this. This innovative treatment has been investigated by leading oncology groups and has yielded successful outcomes and improvement of survival rates. In March 2021, Shorla filed its application for SH-111 with the FDA which was subsequently accepted and granted priority review. We anticipate approval to bring this much needed product to market later this year. We have made a collective effort and worked with various stakeholders to develop SH-111 efficiently and effectively to improve accessibility of a drug that significantly improves outcomes in newly diagnosed patients and mitigate against shortages of the drug product. At Shorla Pharma, the patient’s needs are at the heart of our innovation, and SH-111 is one of our products that will help to improve patient care.
-
 Submission Title: Gene WritingRead More
Submission Title: Gene WritingRead MoreDetails coming soon.
-
 Submission Title: GAVRETO - Rapid Path to FDA ApprovalRead More
Submission Title: GAVRETO - Rapid Path to FDA ApprovalRead MoreBlueprint Medicines is a global precision therapy company that aims to invent life-changing medicines for people with cancer and hematologic disorders. We create therapies that selectively target genetic drivers, with the goal of staying one step ahead across stages of disease. Since its founding just a decade ago, Blueprint Medicines is now delivering two approved medicines – including GAVRETO® (pralsetinib) – directly to patients. GAVRETO is one of nine approved or investigational precision therapies designed by scientists at Blueprint Medicines.
GAVRETO is a kinase inhibitor approved by the FDA for the treatment of three indications: adult patients with metastatic RET fusion-positive non-small cell lung cancer (NSCLC) as detected by an FDA approved test, adult and pediatric patients 12 years of age and older with advanced or metastatic RET-mutant medullary thyroid cancer (MTC) who require systemic therapy, and adult and pediatric patients 12 years of age and older with advanced or metastatic RET fusion-positive thyroid cancer who require systemic therapy and who are radioactive iodine-refractory (if radioactive iodine is appropriate). These indications are approved under accelerated approval based on overall response rate and duration of response. Continued approval for these indications may be contingent upon verification and description of clinical benefit in confirmatory trials.
The RET tyrosine kinase is an oncogenic driver in subsets of patients across multiple tumor types. RET fusions are implicated in approximately 1-2% of patients with NSCLC and approximately 10-20% of patients with papillary thyroid cancer, while RET mutations are implicated in approximately 90% of patients with advanced MTC. In addition, oncogenic RET alterations are observed at low frequencies in colorectal, breast, pancreatic and other cancers. Prior to 2020, therapeutic options for patients with RET-altered cancers were limited.
From 2017 to 2020, Blueprint advanced the development of GAVRETO® (pralsetinib) from clinical trial initiation to FDA approval, representing one of the fastest registration paths for a targeted cancer therapy. Beginning in 2013, the year after the discovery of RET fusions as an oncogenic driver in NSCLC, Blueprint Medicines initiated a research program to design a selective and potent inhibitor of oncogenic RET alterations.
For more information, visit GAVRETO.com.
-
 Submission Title: Development of a New Technology for Obtaining a New Highly Effective Active SubstanceRead More
Submission Title: Development of a New Technology for Obtaining a New Highly Effective Active SubstanceRead MoreEverything is brilliant - simply, on this principle, the action of our drugs is based. They include components that suppress the process of propagation of viral cells and activate the regeneration of healthy liver cells, which first leads to improvement of the condition, and then to complete recovery. Why can MARGALI drugs be trusted? MARGALI offers a number of unique preparations based on plant substances, useful minerals and vitamins.
These are drugs:
• MAC - CIR - for the treatment of all types of hepatitis and cirrhosis of the liver.
• KOHALON - MAC - for the treatment of genital herpes, pyelonephritis, myoma, fibroma, lymphosarcoma and tuberculosis.
Our company's drugs:
• Do not reduce immunity.
• Easy to use.
• Do not cause hallucinations.
• Do not cause deterioration of well-being.
• Do not have any other side effects.Subjective improvement of condition, change of complexion and improvement of appetite are observed after the first week of administration of the drug.
• The size of lymph nodes in the treatment of lymphosarcoma decreases after two to three weeks of taking the drug, and the absolute result is achieved after 6-7 months.
• Liver size in cirrhosis and hepatitis normalizes after two to two and a half weeks of taking the drug. In the treatment of hepatitis B and C, the viral load is reduced by half after 26 days, and the absolute result is achieved after 5-6 months - 78% of patients have a complete absence of hepatitis virus in the blood.
• When treating an open form of tuberculosis of stage 4, it goes into a closed form of stage 3 after 26 days of taking the drug, and the absolute result is achieved after 6-7 months. What is the point? Our drugs not only successfully fight various diseases. They improve the performance of hormonal glands, increase life expectancy and prolong the reproductive period of stem cells. When taking MARGALI drugs, blood is updated every 4 days and the body is gradually rejuvenated. Each of the drugs activates the regeneration of the cells of those organs for the treatment of which it is intended.For example, for cell regeneration: • renal and pulmonary tissue is intended for KOHALON - MAC;
• liver is intended MAC - CIR;
Most Valuable Data & Insights Initiative
This award is for an initiative that harnesses data (internal or RWD), AI, ML or analytics to reveal valuable insights, ultimately resulting in new opportunities for better patient care. We are looking for those who have smartly deployed data or informatics/analysis techniques whether in clinical, commercial, medical or patient-generated data - or who have found ways through data standards or interoperability to enable better insights to be discovered and acted upon.
-
 Submission Title: OneThree Biotech and Jubliant Therapeutics CollaborationRead More
Submission Title: OneThree Biotech and Jubliant Therapeutics CollaborationRead MoreThe initiative being discussed in this submission is one between OneThree Biotech and Jubilant Therapeutics that focuses on the identification of specific biomarkers for new indications in targeted oncology patient populations.
In the collaboration, Jubilant sought to determine the mechanism for its potentially first-in-class dual epigenetic inhibitor targeting melanoma, myelodysplastic syndrome (MDS), and acute myeloid leukemia (AML) and select solid tumor subsets.
Utilizing the OneThree Biotech platform, they were able to:
- Generate new data to support the dual inhibition (LSD1 and HDAC6) to drive inhibition
- Pinpoint molecular markers that could be used to identify patients who might benefit from the dual inhibition
- Determine additional target indications for future trials
OneThree Biotech is a spinout from the largest precision medicine institute in the United States. We have demonstrated great success in drug discovery by applying our biology focused, AI enabled R&D process with partners such as AZ, SPARC, Oncoceutics, and more. In 2021 we began our internal development programs that focus on oncology targets we identified using our AI Platform and validated in early stage development. We won the Reuters Entrepreneur Award in late 2020 and are excited to speak about the results of a partnership we’ve had since then!
Jubilant Therapeutics, Inc. is a patient-centric biopharmaceutical company advancing potent and selective small molecule modulators to address unmet medical needs in oncology and autoimmune diseases. Its advanced discovery engine integrates structure-based design and computational algorithms to discover and develop novel, precision therapeutics against both first-in-class and validated but intractable targets in genetically-defined patient populations. The Company's entrepreneurial minded leadership and scientific teams strive for speed and efficiency by employing a business model that leverages the proven and synergistic capabilities of Jubilant Life Sciences Limited's value chain and shared services. Jubilant Therapeutics is headquartered in the U.S. and guided by globally renowned key opinion leaders and scientific advisory board members. -
 Submission Title: Validating Novel Outcome Measurements - VALINOSRead More
Submission Title: Validating Novel Outcome Measurements - VALINOSRead MoreVALINOS is an abbreviation of Validating Novel Clinical Outcome Measurements - an initiative aiming to advance remote disease assessment.
Using digital tools to enable remote data collection has clear advantages in both clinical practise and in decentralized clinical trials. In a digital world, in-clinic visits can be replaced by remote visits, hence patients need to travel less, while their doctor maintains the clinical oversight remotely. Such a remote approach has become increasingly relevant in the face of the COVID pandemic.
In addition, digital enables more frequent data acquisition, because the patients report data directly to the doctor from his/her own home. The continual data monitoring allows for a better understanding of the treatment and other influencing factors. VALINOS is determined to realise this potential.
Within dermatology, VALINOS validates the outcomes of remote assessments in skin disease by comparing them with in-person assessments by dermatologists. VALINOS currently focuses on psoriasis, atopic dermatitis and actinic keratosis.
The comparative data is collected through a series of clinics, in which multiple dermatologists physically evaluate a patient - to establish a baseline of the patient’s condition. The same patient subsequently uploads photos of their skin to a designated app, which are assessed remotely by a panel of dermatologists. The comparison of the in-clinic assessments and the photo-based assessments form a data set that is used to establish the validity and reliability of the remote method.
All in-clinic evaluations are performed using well-established severity tools specific to a given disease, such as Eczema Area and Severity Index (EASI), Psoriasis Area severity Index (PASI), and Olsen grade for actinic keratosis severity. The remote assessments are based on the same principles and methods as the in-clinic evaluations.
This far, VALINOS has demonstrated a good agreement between the assessments performed remotely and in-clinic. Our findings show excellent reliability of the photo-based remote assessments and clearly indicate that we are on the right track; that remote assessments have the potential to replace and/or support in-clinic visits in dermatology and potentially also other disease areas for the future benefit of patients and doctors alike.
-
 Submission Title: NMQF Clinical Trial Index and Learning CommunityRead More
Submission Title: NMQF Clinical Trial Index and Learning CommunityRead MoreAt Biogen, we are committed to the power of diversity - in our organization and among our patients. Every day we work toward helping to close the access gap in both clinical trials and the healthcare system. The National Minority Quality Forum (NMQF) is a research and educational organization dedicated to reducing patient risk for underrepresented populations, by integrating data and community expertise. Biogen partnered exclusively with NMQF to develop data tools and community level initiatives geared specifically at informing, listening, and designing blueprints to build representative clinical trials.
NMQF built the Biogen-NMQF Clinical Trial Index, a bespoke data and geomapping tool for Biogen comprised of Medicare and Medicaid beneficiary data in Biogen’s disease areas of research. By reporting and displaying real world evidence (RWE) and epidemiologic estimates of disease areas of interest by geography and demography, the Index allows Biogen to be more strategic with identifying the right sites in the right locations to bring clinical trials to the communities we need to serve.
Based on the Index data, NMQF and Biogen are building a Clinical Trial Learning Community (CTLC). The CTLC is a virtual space where local investigator sites can interact with local providers, community leaders, and industry towards a common objective: reduce disparities in care and outcomes for underrepresented patient populations by ensuring awareness of and access to available clinical trials close to home.
The novel Biogen-NMQF Clinical Trial Index leverages real-world evidence and population data to improve diversity in all aspects of clinical trial planning, recruitment, and execution. The CTLC enables clinical trial leaders to develop an action plan and blueprint for successful site and patient engagement in clinical trials at the community level including site selection, outreach, education, access & referral.
-
 Submission Title: Trial Pathfinder: Using Machine Learning to Advance Inclusive ResearchRead More
Submission Title: Trial Pathfinder: Using Machine Learning to Advance Inclusive ResearchRead MoreTrial Pathfinder, created in partnership between Genentech and the Stanford University Institute for Human-Centered Artificial Intelligence, uses real-world data (RWD) and artificial intelligence (AI) to evaluate and predict eligibility criteria’s impact on clinical trials. Incorporating RWD can potentially lead to more inclusive trials, while reducing time and costs needed to bring new medicines to patients faster.
Specifically, through Trial Pathfinder we emulated 10 completed lung cancer trials and showed we could increase the pool of eligible patients by 107% on average -- including expanding inclusion of women, African-Americans, and a broader range of ages -- while maintaining treatment efficacy and patient safety.
The team leveraged RWD from 61,094 individuals with advanced non-small cell lung cancer from the nationwide Flatiron Health electronic health record-derived database. Trial emulation included selecting individuals who met the available eligibility criteria as originally published in clinical trial protocols. The team developed a computational pipeline which optimised data-driven eligibility criteria that would benefit the most patients by systematically emulating thousands of trials with varying combinations of eligibility criteria.
Survival analysis was performed using the hazard ratio of overall survival as the outcome. A novel implementation of the Shapley value was created to provide a principled way to evaluate each eligibility criterion’s effect on the resulting hazard ratio across the thousands of experiments.
Trial Pathfinder demonstrated it can help select an optimal subset of eligibility criteria that consistently maintains or reduces the hazard ratio, while maximising the pool of eligible patients. Additionally, since Trial Pathfinder is open-source, the scope of applications will hopefully expand to other types of cancer, and potentially other diseases.
The emergence and combination of quality RWD and advanced technologies, including AI and machine learning, present new opportunities to accelerate R&D, bring effective medicines to the right people quickly, and potentially reduce the burden on healthcare systems. This is why projects like Trial Pathfinder are central to realising the future of personalised healthcare.
-
 Submission Title: ImmunoLab - Sanofi Immunology Data Discovery LabRead More
Submission Title: ImmunoLab - Sanofi Immunology Data Discovery LabRead MoreThe Sanofi Immunology Data Discovery Lab (“ImmunoLab”) is a transformative platform which maximizes the use of digitized healthcare data to standardize and enable rapid generation of robust insights. It enables understanding of the dynamic patient need in the real-world, all at the user’s fingertips. By changing how we use real world data, we aim to improve patient outcomes through optimized management and care.
With the increasing availability of digitized healthcare data and broadening demand for RWE across pharma, scalable platforms like ImmunoLab are a required alternative to the current one-question-at-a-time approach for generating evidence.
ImmunoLab uses an intuitive interface and applies artificial intelligence to analyze an Immunology and Inflammatory Disease Patient Cohort from the US Optum Electronic Health Record. The platform facilitates real-world hypothesis generation across a range of diseases. Optum data enables Sanofi teams and external partners to analyze patient data from over 16 million patients. The patient cohort includes over 30 inflammatory diseases (and related comorbidities) with more than 1,500 features available per patient, spanning a 5-year time-frame. This expansive data-set enables over 83 million analyses that may explored through three analytical modules.
In each user-centric module, inclusion criteria and input variables can be easily modified and are analyzed by machine learning algorithms. The analyses outcomes are presented in intuitive visual formats. The three modules include the Patient Journey Mapper which describes patient characteristics and drug treatment patterns, the Switch Modeler which analyzes treatment switching behavior and its drivers, and the Head-to-Head Simulator which models comparative effectiveness of treatments.
ImmunoLab can help shape the way that life sciences companies generate insights, innovating from traditional approaches to more scalable tools. We have seen how research is evolving and are proud to be identifying faster routes to understand how diseases might be predicted, prevented, or cured. To turn the analysis of patient journeys into improved patient care is the ultimate goal of ImmunoLab.
Most Valuable Education Initiative
This award is for an initiative that has raised the profile and visibility of a disease, cause, health concern, population or treatment. We are not looking to reward mere advertising (no matter how good it is); we are seeking campaigns which have provided additional value, shown creative ways to raise awareness, gathered further reach in an additional way, clarified scientific concepts for HCPs or patients, or made things easier or clearer.
-
 Submission Title: Pediatric IBD: A Growing ConcernRead More
Submission Title: Pediatric IBD: A Growing ConcernRead MoreThe “Speaking from Experience” series is planned to be an expansive series of interactive and emotional patient stories that connect HCPs to the real impacts—physical, mental, and emotional—of patients suffering from autoimmune diseases like inflammatory bowel disease (IBD). In this first entry, “Pediatric IBD: A Growing Concern”, the initiative tackles the issue of children who are diagnosed with IBD in their young and adolescent years. Addressing IBD is a lifelong battle, which means children who go undiagnosed or untreated face potentially significant disease burden throughout their lives. This initiative targets HCPs who may encounter young or adolescent patients with IBD, such as pediatricians and gastroenterologists, and gives them a first-hand look at the diagnosis and treatment process. IBD is a life-changing diagnosis that requires a comprehensive disease management approach; it requires potential lifestyle changes, mental and emotional health support, and a strong, connected network of HCPs and caretakers working together to help improve patient outcomes. https://www.tipmedia.com/demos/PedsBrandonHall
-
 Submission Title: The Facial Anatomy ApplicationRead More
Submission Title: The Facial Anatomy ApplicationRead MoreDetails coming soon.
-
 Submission Title: CIN Risk Disease AwarenessRead More
Submission Title: CIN Risk Disease AwarenessRead MoreSometimes you must agitate to educate. This is the thinking behind the CIN Risk campaign developed by Proximyl Health for BeyondSpring. Their breakthrough therapy, plinabulin, is approaching launch for the prevention of chemotherapy-induced neutropenia. However, many oncologists falsely believe that they have CIN under control with the standard of care, G-CSFs, which do not take full effect for the first seven days of chemo. This misperception does not acknowledge the frequent infections that can delay a patient’s chemo regimen, reduce the dose, or even cause hospitalization—all impacting survival outcomes.
Branding a medical condition requires capturing the menacing force confronting both HCPs and patients in the most dramatic way. To raise awareness of the vital risks that need not be accepted as part of chemotherapy, Proximyl Health had to raise the stakes. The agency boldly executed a metaphoric concept designed to get HCPs to sit up and take notice. In effect, it established the “villain” that plinabulin could vanquish.
Since Proximyl Health has unmatched access to HCPs and patients, it conducted its unique Living Creative process—a session where concepts are co-created directly with the customer segments to arrive at ideas that are pre-vetted for impact and engagement.
The result: an attention-grabbing campaign dramatizing the ever-present threat of CIN, especially during the first cycle of chemotherapy. The nucleus of the initiative is CINrisk.com—a website specifically created to mandate a revised level of concern and response. The visual features a chemo bag’s access line turning into a vicious snake, ready to prey on the patient’s immune system with needle-like fangs. The headline—CIN can strike at any moment—drives home the impactful effect of the animation. Users can also find an interactive quiz that engages their participation. In addition to the website, the full campaign rollout includes a journal ad, advertorial, emails, KOL videos, and virtual exhibit booths at SABCS and ASCO. Taken together, the campaign earned 45,147,947 impressions from paid media, 871,749 social media engagements, and 238,710 users on the website and virtual booth—all evidence that Proximyl Health can create critical mass for any client challenge.
-
 Submission Title: The Accompany Project - Standardization of Diagnosis and Treatment in PSCI (China)Read More
Submission Title: The Accompany Project - Standardization of Diagnosis and Treatment in PSCI (China)Read MoreExecutive Summary
PSCI (Post-stroke cognitive impairment) is much common in stroke patients leading to huge disease burden. However, the diagnosis and treatment of PSCI in China is so poor and worth serious consideration.The project mainly includes three modules:
HCPs (healthcare providers) education: based on the 2021 Chinese PSCI Expert Consensus, the project supports national experts to practice education activities for grassroots HCPs to improve their awareness and capability on PSCI standard diagnosis and treatment. Up to now, more than 20 meetings were held nationwide and covering more than 600 neurologists.Patient education: education meetings were held in the neurology ward for patients with acute stroke and their caregivers before discharge when they have the strongest demand for disease management. It can greatly improve patients' participation and learning effect. Patient education meetings also held more than 20 sessions, covering nearly 400 acute stroke patients.
Intelligent tool on cognitive assessment scale: The applet sets up patient and doctor evaluation’s ports including commonly used scales with voice reading and automatic scoring. The tool increased the usage of the scales and improved the feasibility of doctors to standardize the diagnosis of PSCI in a digital way.
It is worth mentioning that the tool is equipped with the localized MoCA (Montreal Cognitive Assessment) scale newly revised by Chinese PSCI experts. Via the project data, the reliability and validity of the localized scale will be verified. In addition, the experts are gratifying to find out the cut-off point of the MoCA scale might be redefined. It would provide the new starting point for the effective implementation of PSCI standardized diagnosis and treatment in China.
These results have proved the educational significance of the project, which is not only for doctors to learn disease knowledge, but also to provide a platform for using the knowledge. And the practice achievement will also be fed back to standardize the diagnosis and treatment education.
-
 Submission Title: Rethink Fabry™Read More
Submission Title: Rethink Fabry™Read MoreChiesi Global Rare Diseases launched the Rethink Fabry™ education program to build broader awareness of Fabry disease and provide patients, caregivers and healthcare professionals with new resources and insights to help them make optimal decisions. Rethink Fabry is designed to put a spotlight on the significant impact Fabry disease has on the daily lives of patients and families. It also aims to highlight the continuing areas of unmet medical need in patient care and strategies to shorten time to diagnosis.
Through a special webinar and video series, Rethink Fabry has made perspectives from leading experts dedicated to improving the current standard of care accessible by all members of the Fabry disease community. Several renowned specialists who have spent their careers caring for patients living with Fabry disease have contributed to the video series to share insights on a range of important topics such as the psychosocial impact of the disease, the importance of proactive treatment and what the future holds for the care of Fabry. With ongoing support from leaders in the medical community, Rethink Fabry is positioned to connect patients and healthcare providers with expert advice and information.
The Rethink Fabry website serves as a hub for new resources created with patients and healthcare providers in mind with the goal of building a brighter future for the Fabry disease community. Doctor dialogue guides, daily symptom tracker checklists and annual monitoring guides are just a few of the resources Rethink Fabry provides to support patients and healthcare providers navigating the diagnosis and treatment of Fabry disease.
As with any rare disease, working to build broader awareness of Fabry disease and delivering patients, caregivers and healthcare providers the information they need to make informed decisions is an important and ongoing need. Chiesi Global Rare Diseases is deeply committed to working with all members of the Fabry disease community to achieve this goal. Through Rethink Fabry, Chiesi Global Rare Diseases hopes to bring new levels of support to everyone affected by Fabry disease.
The Accompany Project - Standardization of Diagnosis and Treatment in PSCI (China), Viatris
PSCI (Post-stroke cognitive impairment) is much common in stroke patients leading to huge disease burden. However, the diagnosis and treatment of PSCI in China is so poor and worth serious consideration. The Accompany Project-Standardization of Diagnosis and Treatment in PSCI is jointly initiated by the Vascular Cognitive Impairment Branch, the Chinese Stroke Association and sponsored by Medical Affairs & Clinical Research department, Greater China Region, Viatris.
The project mainly includes three modules:
HCPs (healthcare providers) education: based on the 2021 Chinese PSCI Expert Consensus, the project supports national experts to practice education activities for grassroots HCPs to improve their awareness and capability on PSCI standard diagnosis and treatment. Up to now, more than 20 meetings were held nationwide and covering more than 600 neurologists.
Patient education: education meetings were held in the neurology ward for patients with acute stroke and their caregivers before discharge when they have the strongest demand for disease management. It can greatly improve patients' participation and learning effect. Patient education meetings also held more than 20 sessions, covering nearly 400 acute stroke patients.
Intelligent tool on cognitive assessment scale: The applet sets up patient and doctor evaluation’s ports including commonly used scales with voice reading and automatic scoring. The tool increased the usage of the scales and improved the feasibility of doctors to standardize the diagnosis of PSCI in a digital way.
It is worth mentioning that the tool is equipped with the localized MoCA (Montreal Cognitive Assessment) scale newly revised by Chinese PSCI experts. Via the project data, the reliability and validity of the localized scale will be verified. In addition, the experts are gratifying to find out the cut-off point of the MoCA scale might be redefined. It would provide the new starting point for the effective implementation of PSCI standardized diagnosis and treatment in China.
These results have proved the educational significance of the project, which is not only for doctors to learn disease knowledge, but also to provide a platform for using the knowledge. And the practice achievement will also be fed back to standardize the diagnosis and treatment education.
Most Valuable Awareness Initiative
This award is for an initiative that has raised the profile and visibility of a disease, cause, health concern, population or treatment. We are not looking to reward mere advertising (no matter how good it is); we are seeking campaigns which have provided additional value, shown creative ways to raise awareness, gathered further reach in an additional way, clarified scientific concepts for HCPs or patients, or made things easier or clearer.
-
 Submission Title: Making Scientific Research Available to Prostate Cancer Patients & Caregivers in an Accessible and Understandable Format to Inform and Educate, BayerRead More
Submission Title: Making Scientific Research Available to Prostate Cancer Patients & Caregivers in an Accessible and Understandable Format to Inform and Educate, BayerRead MorePlain Language Summary (PLS) is a description consisting of text and/or images, which conveys complex information in a simple, easy to understand fashion and is used by a general audience without a medical or research background. While the concept of plain language summary publications is still in its infancy in the medical publishing industry, Bayer's unique and innovative approach to the development of various PLS publications has focused on having the patient voice at the forefront and optimizing patient education and their understanding of relevant trial results.
In order to make scientific information available to prostate cancer patients, Bayer partnered with patients to publish the trial results of a Phase III prostate cancer study called ARAMIS. This peer-reviewed, patient-authored publication was published in Future Oncology with full open access along with an animated video. Following the success of this publication, we are continuing to prioritize patient focused publications with digital enhancements and offer opportunities to involve patient advocates to present their perspectives. We have shared insights on the various formats of Plain Language Summary (PLS) of scientific publications and examples of communication best practice to improve patient experience throughout their cancer journey at Drug Information Association (DIA) Global Annual Meeting (June 27 – July 1, 2021) and European Association of Urology (EAU) 36th Annual Meeting (July 9, 2021), respectively. Accompanying digital elements (video abstracts) have also been made available highlighting the key points in these patient focused presentations.
In summary, developing and publishing diverse formats of PLS in open access venues can extend the reach of scientific data to broad audiences, while at the same time, creating transparency and equity in healthcare treatment and shared decision making. We intend to scale this initiative across the Bayer portfolio in collaboration with patients and patient advocates.
-
 Submission Title: #RAREis Uplifting the Rare Disease CommunityRead More
Submission Title: #RAREis Uplifting the Rare Disease CommunityRead More#RAREis began as social media campaign launched by Horizon Therapeutics in 2017 to elevate the voices, faces and experiences of the rare disease community. It has since grown into a multi-faceted program that connects people living with rare diseases and their families to resources that support, inform and educate them as they navigate their daily lives. The Power of Storytelling #RAREis brings rare disease stories to life. By elevating personal stories of living with a rare disease, #RAREis empowers individuals to share the uncensored truth of their journey. It has given a platform and a voice to those living with diseases so rare that no dedicated organization exists for their condition.
#RAREis is anchored by a community website as well as an Instagram and Facebook page. Individuals are encouraged to create their own #RAREis photo and share their story via social media. The #RAREis hashtag (#) is a way to follow the conversation and remains in the name and logo to represent the broader movement and community. #RAREis Blog features the personal stories of individuals, families, researchers, physicians and rare disease advocates to bring together firsthand experiences that are common in the lives of those impacted by rare diseases.
#RAREis Resources program helps individuals and families impacted by rare disease understand the obstacles they might encounter during various life transitions or while dealing with difficult topics like genetic testing. #RAREis provides resources that strengthen health literacy and disease management among the rare disease community, and connects them to organizations that offer supportive programs and services.By bringing together the rare disease community, #RAREis generates awareness, support and education for hundreds of rare disease states. #RAREis Voices is a video series featuring leading physicians, researchers, advocates, individuals and parents who share common experiences faced by many people in the rare disease community. RAREisCommunity.com A website that connects people living with rare diseases and their families to unique resources developed by and f for the rare disease community, personal stories and links to advocacy partners to stay connected.
-
 Submission Title: Carer Well-Being Index: Recognizing and Providing Support for Unpaid CarersRead More
Submission Title: Carer Well-Being Index: Recognizing and Providing Support for Unpaid CarersRead MoreAs a healthcare provider, Merck KGaA, Darmstadt, Germany has always been acutely aware of the critical role family carers, often referred to more inclusively as unpaid carers, play in the patient journey. The International Alliance of Carer Organization (IACO)’s 2021 Global State of Caring estimates that there are 259 million of them. And even at the best of times, carers suffer tremendous stress. With COVID-19, there was the addition of extreme isolation and heightened logistical and financial hardships.
It was time for everyone—private and public sectors alike—to step up and recognize the contribution carers make to our society, and to give them the support they need. Embracing Carers®, led by Merck KGaA, Darmstadt, Germany in collaboration with nine leading carer organizations, set out to quantify, for the first time, the global impact of the pandemic on carers. The goal: to spur both private and public sector awareness and insight into the plight of unpaid carers that would lead to direct and dramatic action by parties across the governmental, NGO and private-sector spectrum.
By surveying more than 9,000 unpaid carers across 12 countries and four continents, Merck KGaA, Darmstadt, Germany identified five key trends related to unpaid carers. Known as the Carer Well-Being Index, it is the first-of-its-kind study focusing on the impact of the pandemic on unpaid carers’ holistic well-being, including their emotional, physical and financial health. The survey was met with extraordinary enthusiasm and resulted in extensive coverage in top-tier media outlets and academic publications, garnering more than 300 pieces of coverage and more than 830 million estimated impressions.
By identifying carers’ current state, Merck KGaA, Darmstadt, Germany was able to recommend specific actions to help governments and private sectors in these countries alike take immediate action. The survey also spurred action in the form of new research and new proposed legislation to close gaps in support by focused measures from multiple players. And as we look to the future, Embracing Carers® plans to continue driving recognition, access to resources, visibility among policymakers and innovative new collaborations with other organizations to ease the burden on unpaid carers.
-
 Submission Title: New Normal, Same CancerRead More
Submission Title: New Normal, Same CancerRead MoreBy the third quarter of 2020, cancer diagnoses had fallen by approximately 40%. A fear of contracting COVID-19 at hospitals and clinics was deterring patients across the world from seeking medical attention for new symptoms or attending routine checks.
AstraZeneca wanted to create a fully integrated creative campaign aimed at raising awareness amongst policymakers, HCPs, patient advocacy groups (PAGs), and the public to encourage people with cancer to resume treatment, if paused, and those with symptoms to consult a doctor and get checked.
The world may have been adapting to a new normal, but cancer remained the same and continued to be a global challenge. Our New Normal, Same Cancer (NNSC) campaign was designed to juxtapose COVID-related scenes with messages around seeking, or resuming, cancer treatment. The campaign delivered a simple message, which worked for policy and consumer behaviour change: Don’t wait. Contact your doctor. Get checked. and took just four weeks from realisation to activation in October 2020.
Since the global roll-out of NNSC on 1 October 2020, it has now launched in 67 countries and achieved several “industry firsts”, including government endorsement of the campaign (a first for the pharma industry in Australia), roll-out of the first national children’s cancer PAG partnership in Lebanon and the first in-store grocery advertising in The Netherlands. The campaign went to new lengths to reach people wherever they are.
Using social media as the focus of communication, the campaign slogan reached more than 1.5 billion people across the world.
Our strategy was to:
• Unite patients and caregivers to overcome fear of visiting hospitals and clinics
• Overcome barriers to the access of treatment
• Collaborate with a patient advocacy group (PAG) steering committee to ensure that all messaging, visuals and materials would resonate with those in the best position to reverse the impact seen
• Leverage AstraZeneca’s existing platforms and contacts
• Leverage local markets’ in-depth knowledge of the COVID-19 situation at a national level and give local teams the autonomy to launch at the right time
• Integrate key policy and consumer behavioural insights on how the public respond to public health asks and patient insight -
 Submission Title: Discovering DiabetesRead More
Submission Title: Discovering DiabetesRead MoreDiabetes is a silent but deadly killer.
As per the International Diabetes Federation (IDF), Pakistan has over 19.4 million diabetic patients, and the number is likely to increase to 37.1 million by 2045 – a truly horrifying scenario. This is alarming because statistically, there is 1 diabetes specialist for every 16,166 patients and 1 Diabetes Center for every 718,518 patients.
PharmEvo with its dream of promoting ‘a healthier society’ decided to tackle this problem by partnering with the Pakistan Endocrine Society to establish a breakthrough program ‘Discovering Diabetes’ (DD). This program aims to raise awareness of diabetes, track high-risk diabetic patients across Pakistan, and connect them to qualified healthcare professionals for the right treatment.
Discovering Diabetes raises awareness through a 3-step approach i.e., track, connect and serve. The Discovering Diabetes team tracks diabetic and pre-diabetic patients through various physical camps and social media drives, and then administer free risk assessment questionnaires and diagnostic testing. Till now 250,000 People have been screened & 200 Discovering Diabetes clinics have been launched since the start of this project in March 2021.After risk assessment, the at-risk patients are then contacted by an IDF Certified Diabetes Educator from PharmEvo to provide disease management information. The patients are then directed to the free teleconsultation service, where a qualified healthcare physician may further refer them to our diagnostic partner, Chughtai Lab., for a free blood sugar screening and HbA1c test. Based on the results, the patient will be referred to get free consultation from a partner Health Care Professional.
Through this cycle, Discovering Diabetes ensures that the patient is facilitated throughout the process. Moreover, a National diabetes prevalence study is also planned at the Holy Family Hospital, Rawalpindi, under the supervision of Dr. Madiha Nazar.
Additionally, patients are provided information via the website, regular social media awareness messages, helpful articles by various healthcare professional published in Daily Jung, our cause-partners. For TV, DD awareness programs were run on Morning shows, along with social drives by celebrities, such as Imran Abbas and Waseem Badami. On-ground awareness activities included posters and wobblers placed in 10,000 different locations.


